Molecular orbital diagram of Li2 & Be2: Number of electrons in Li2 molecule =6 Answer to Draw an MO energy diagram and predict the bond order of Be2+ and ...
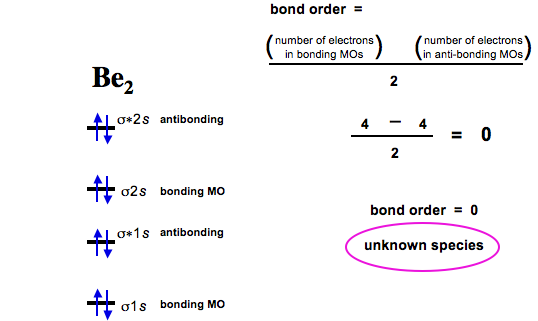
(ii) (iii) Use molecular orbital theory to explain why Be2 molecule does not exist? ... (σ2s)2, (σ*2s)2 Molecular Orbital Diagram: Bond order = Nb −Na2 = 4− 4=0 ...
Be2 e.. O2.. 2+.. 2.. Refer to Diagram 9-1.. According to molecular orbital theory, which of the following species is least likely to exist.. (i.e., has the lowest bond order)?.
Gay picture Be2 Lewis Structure, find more lesbian how to draw the be lewis dot structure youtube, be mo diagram wiring diagram, b lewis structure how to draw ...
Mar 20, 2020 — ... to 1, which you can get by drawing the molecular orbital diagram and performing the ... Your question is wrong because be2 molecule does not exist as be2 ... This pairs up with the electron in the highest occupied σ-orbital.
Dec 15, 2020 — 1 Answer to li2 is stable but be2 is not stable .. explain, molecular orbital diagram.
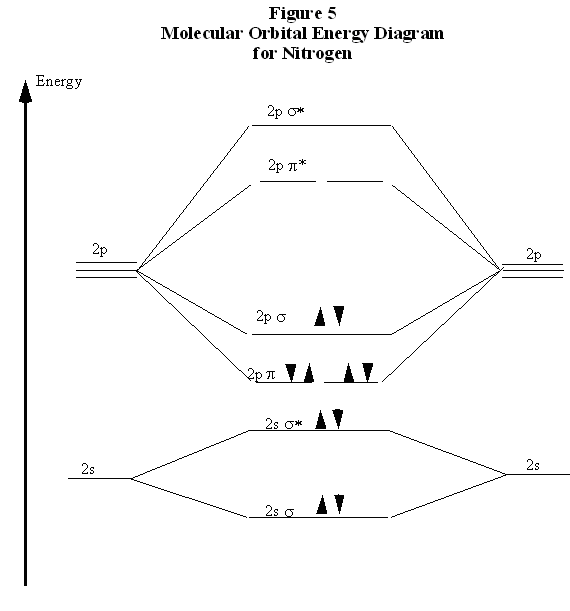
shown on the diagram, ..
Be2. how-to-open-graco-swing-motor
molecular orbital diagram
(1σ)2(1σ*)2.. ½ (2 – 2) = 0.. (no bond).. Be2.. +.. (1σ)2(1σ*)1.. ½ (2 – 1) = ½ decrease.. Be2.. − ... has an electron in the σ bonding orbital formed from overlap of the 3s orbitals. tamilrockers_secret_superstar_with_subtitle_torrent_file_molecular orbital diagram of o2

... enough to hold the molecule together.
Molecular Orbitals of Li₂ to F₂ This video shows the end of the Be2 molecule MO diagram and explains pi orbitals, paramagnetism, and the MO diagrams for ...
Thus we can draw ENERGY LEVEL DIAGRAM for m.o.'s of H2.. : 1sA ... "Be2.. " σs.. 2σ*s.. 2.. Bond order = 0 - no net bonding energy, so molecule does not exist.
MO Energy Level Diagram For Homonuclear Diatomic Molecules of Second ... Be2 Molecule is not stable as stabilization energy is 0 and bond order is 0.. ▫.
14 hours ago — For the ion Be2+ : a) Draw the molecular orbital diagram .. b) Calculate the bond order.. c) Would this ion exist? d) Write the electron ... 8 years ...
(a) To what does a zero energy correspond in the diagram? (b) According to ... (b) According to molecular orbital theory, would Be2 or Be2.. + be more stable?
Would the answer be Be2 because it has the most empty p orbitals? .. Лето в лагере, P9143474 @iMGSRC.RU
3e88dbd8be

